trending topics
market reports
-

Registration Now Open: MEDICAL JAPAN 2026 OSAKA – Western Japan’s Largest Healthcare Trade Show
2026-02-10
-

MEDICAL JAPAN 2025 OSAKA Returns to Showcase Global Innovations
2025-02-17
-

Visit MEDICAL JAPAN 2023 TOKYO and take full advantage of the business opportunities!
2023-09-01
-
US to distribute 400 million free N95 masks at CVS, Walgreens in COVID fight
2022-01-21
-

Ethiopia receives additional 2.2 mln doses of Chinese-donated COVID-19 vaccines
2022-01-21
-
Hong Kong researchers say they develop novel material able to kill COVID-19 virus
2022-01-14
-

10 million more Chinese doses on way for Kenya
2022-01-14
-

Sino-African ties on track for a brighter future
2022-01-07
-
Efforts urged to boost COVID-19 vaccine production capacity in poor countries
2022-01-07
-
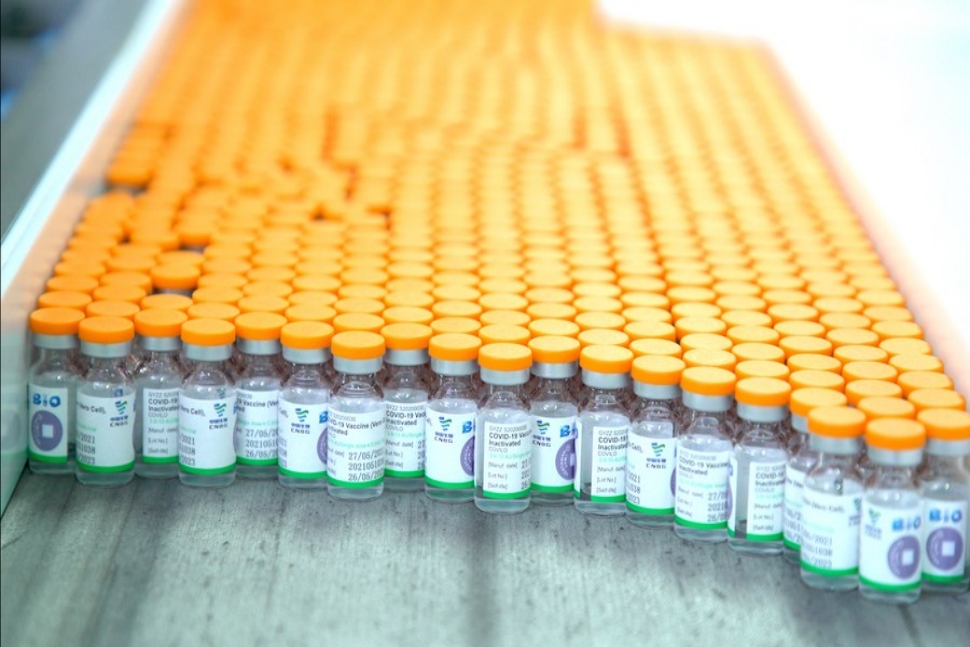
UAE approves Sinopharm's new protein-based COVID-19 vaccine
2022-01-07
UAE approves Sinopharm's new protein-based COVID-19 vaccine
2022-01-07
Photo taken on June 1, 2021 shows vials of the Sinopharm vaccine in Beijing, capital of China. [Photo/Xinhua]
DUBAI - The United Arab Emirates (UAE) has approved the emergency use of a new recombinant protein vaccine manufactured by China's Sinopharm, the health ministry said on Tuesday.
In a statement published on its official website, the ministry said "the emergency use of the new vaccine is in full compliance with the regulations and laws for a faster review of licensing procedures".
The UAE health authority said the protein-based Sinopharm vaccine was approved after strict monitoring and evaluation of the data of the study conducted in the UAE, which included individuals who were previously vaccinated with two doses of the Sinopharm inactivated vaccine.
The protein-based vaccine will be used as a booster jab in the UAE, which has fully vaccinated over 91 percent of its population.
The new Chinese vaccine demonstrated an improved immune capacity against the new variants, "with a high safety rate that allows rapid production and easy storage and distribution," the ministry added.
The UAE approved the Sinopharm inactivated vaccine on Dec 9, 2020, to boost the country's vaccination drive against the virus.
(Xinhua)



 My Member
My Member Message Center
Message Center











